Marine Drug goes to Phase III trials to treat Lung Cancer
04/08/2016
PharmaMar is starting a new Phase III trial, with patients suffering from small cell lung cancer (SCLC). It’ll test the efficacy of its third candidate obtained from marine organisms (PM1183) as a SCLC treatment.
Now, PM1183 is under the spotlight. This cancer drug will be used in a second Phase III trial (after ovarian cancer). It’ll enrol 600 patients with small cell lung cancer (SCLC), for whomplatinum-based treatments have failed before.
This type of lung cancer is less common than non-small cell lung cancer (which has very bad statistics) but has the worstprognosis for pulmonary cancers. Five years after diagnosis, only 5% to 10% of the patients are still alive.

Small cell lung cancer micrograph. (CC 3.0 Michael Bonert/Nephron)
The trial will evaluate the efficacy of PM1183 as a therapy, in combination withdoxorubicin (chemotherapy drug). The therapy’s effect on the progression-free-survival (PFS) of patients will be compared to those undergoing to other treatments – a chemotherapy combination (cyclophosphamide, doxorubicin and vincristine) or topotecan.
Topotecan is currently the only drug approved for this condition, both in the US and in Europe. This lack of therapyoptions also contributes to the poor outlook of small cell lung cancer. If successful, PM1183 would be able to cover a high unmet medical need.
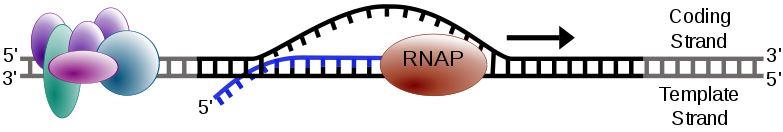
PM1183 inhibits over-active DNA transcription in solid cancers. (CC 0 Forluvoft)
PharmaMar’s PM1183 (also known aslurbinectedin) acts as an inhibitor of RNA polymerase II, an enzyme involved in thetranscription of DNA. In certain types of tumor, the transcription is over-activated – a process where this enzyme is essential.
As with PharmaMar’s other candidates, this drug is the result of a research program to find marine compounds with antitumor activity. The strategy already yielded Yondelis, approved to treat soft tissue sarcoma and ovarian cancer.